4 results
Colonization with extended-spectrum cephalosporin-resistant Enterobacterales (ESCrE) in hospitalized patients in Botswana
- Naledi Betsi Mannathoko, Mosepele Mosepele, Robert Gross, Rachel Smith, Ashley Styczynski, Leigh Cressman, Melissa Richard-Greenblatt, Laurel Glaser, Kevin Alby, Anne Jaskowiak, Kgotlaetsile Sewawa, Laura Cowden, Dimpho Otukile, Giacomo Paganotti, Margaret Mokomane, Warren Bilker, Ebbing Lautenbach
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s81
-
- Article
-
- You have access Access
- Open access
- Export citation
-
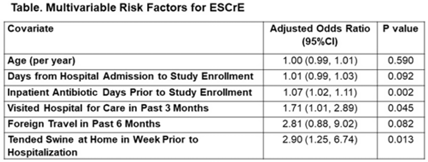
Background: The epidemiology of extended-spectrum cephalosporin-resistant Enterobacterales (ESCrE) in hospitalized patients in low- and middle-income countries (LMICs) is poorly described. Although risk factors for ESCrE clinical infection have been studied, little is known of the epidemiology of ESCrE colonization. Identifying risk factors for ESCrE colonization, which can predispose to infection, is therefore critical to inform antibiotic resistance reduction strategies. Methods: This study was conducted in 3 hospitals located in 3 districts in Botswana. In each hospital, we conducted ongoing surveillance in sequential units hospitalwide. All participants had rectal swabs collected which were inoculated onto chromogenic media followed by confirmatory testing using MALDI-TOF MS and VITEK-2. Data were collected via interview and review of the inpatient medical record on demographics, comorbidities, antibiotic use, healthcare exposures, invasive procedures, travel, animal contact, and food consumption. Participants with ESCrE colonization (cases) were compared to noncolonized participants (controls) using bivariable and multivariable analyses to identify risk factors for ESCrE colonization. Results: Enrollment occurred from January 15, 2020, to September 4, 2020, and 469 participants were enrolled. The median age was 42 years (IQR, 31–58) and 320 (68.2%) were female. The median time from hospital admission to date of sampling was 5 days (IQR, 3–12). There were 179 cases and 290 controls (ie, 38.2% of participants were ESCrE colonized). Independent risk factors for ESCrE colonization were a greater number of days on antibiotic, recent healthcare exposure, and tending swine prior to hospitalization. (Table). Conclusions: ESCrE colonization among hospitalized patients was common and was associated with several exposures. Our results suggest prior healthcare exposure may be important in driving ESCrE. The strong link to recent antibiotic use highlights the potential role of antibiotic stewardship interventions for prevention. The association with tending swine suggests that animal husbandry practices may play a role in community exposures, resulting in colonization detected at the time of hospital admission. These findings will help to inform future studies assessing strategies to curb further emergence of hospital ESCrE in LMICs.
Disclosures: None
Validation of an electronic algorithm to identify appropriate antibiotic use for community-acquired pneumonia in children
- Kathleen Chiotos, Robert Grundmeier, Didien Meyahnwi, Lauren Dutcher, Ebbing Lautenbach, Melinda Neuhauser, Keith Hamilton, Anne Jaskowiak, Leigh Cressman, Julia Szymczak, Brandi Muller, Jeffrey Gerbe
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s106
-
- Article
-
- You have access Access
- Open access
- Export citation
-
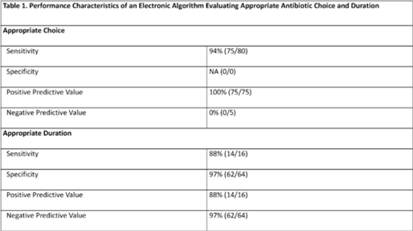
Background: Community-acquired pneumonia (CAP) is a common indication for antibiotic use in hospitalized children and is a key target for pediatric antimicrobial stewardship programs (ASPs). Building upon prior work, we developed and refined an electronic algorithm to identify children hospitalized with CAP and to evaluate the appropriateness of initial antibiotic choice and duration. Methods: We performed a cross-sectional study including children 6 months to 17 years hospitalized for CAP between January 1, 2019, and October 31, 2022, at a tertiary-care children’s hospital. CAP was defined electronically as an International Classification of Disease, Tenth Revision (ICD-10) code for pneumonia, a chest radiograph or chest computed tomography scan (CT) performed within 48 hours of admission, and systemic antibiotics administered within the first 48 hours of hospitalization and continued for at least 2 days. We applied the following exclusion criteria: patients transferred from another healthcare setting, those who died within 48 hours of hospitalization, children with complex chronic conditions, and those with intensive care unit stays >48 hours. Criteria for appropriate antibiotic choice and duration were defined based on established guidelines. Two physicians performed independent medical record reviews of 80 randomly selected patients (10% sample) to evaluate the performance of the electronic algorithm in (1) identifying patients treated for clinician-diagnosed CAP and (2) classifying antibiotic choice and duration as appropriate. A third physician resolved discrepancies. The electronic algorithm was compared to this medical record review, which served as the reference standard. Results: Of 80 children identified by the electronic algorithm, 79 (99%) were diagnosed with CAP based on medical record review. Antibiotic use was classified as the appropriate choice in 75 (94%) of 80 cases, and appropriate duration in 16 (20%) of 80 cases. The sensitivity of the electronic algorithm for identifying appropriate initial antibiotic choice was 94%; specificity could not be calculated because no events of inappropriate antibiotic choice were identified based on chart review. The sensitivity and specificity for determining appropriate duration were 88% and 97%, respectively (Table 1).
Conclusions: The electronic algorithm accurately identified children hospitalized with CAP and demonstrated acceptable performance for identifying appropriate antibiotic choice and duration. Use of this electronic algorithm may improve the efficiency of stewardship activities and could facilitate alignment with updated accreditation standards. Future studies validating this algorithm at other centers are needed.
Disclosures: None
Development of an Electronic Algorithm to Identify Inappropriate Antibiotic Prescribing for Pediatric Pharyngitis
- Jeffrey Gerber, Robert Grundmeier, Keith Hamilton, Lauri Hicks, Melinda Neuhauser, Nicole Frager, Muida Menon, Ellen Kratz, Anne Jaskowiak, Leigh Cressman, Tony James, Jacqueline Omorogbe, Ebbing Lautenbach
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s188-s189
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Antibiotic overuse contributes to antibiotic resistance and unnecessary adverse drug effects. Antibiotic stewardship interventions have primarily focused on acute-care settings. Most antibiotic use, however, occurs in outpatients with acute respiratory tract infections such as pharyngitis. The electronic health record (EHR) might provide an effective and efficient tool for outpatient antibiotic stewardship. We aimed to develop and validate an electronic algorithm to identify inappropriate antibiotic use for pediatric outpatients with pharyngitis. Methods: This study was conducted within the Children’s Hospital of Philadelphia (CHOP) Care Network, including 31 pediatric primary care practices and 3 urgent care centers with a shared EHR serving >250,000 children. We used International Classification of Diseases, Tenth Revision (ICD-10) codes to identify encounters for pharyngitis at any CHOP practice from March 15, 2017, to March 14, 2018, excluding those with concurrent infections (eg, otitis media, sinusitis), immunocompromising conditions, or other comorbidities that might influence the need for antibiotics. We randomly selected 450 features for detailed chart abstraction assessing patient demographics as well as practice and prescriber characteristics. Appropriateness of antibiotic use based on chart review served as the gold standard for evaluating the electronic algorithm. Criteria for appropriate use included streptococcal testing, use of penicillin or amoxicillin (absent β-lactam allergy), and a 10-day duration of therapy. Results: In 450 patients, the median age was 8.4 years (IQR, 5.5–9.0) and 54% were women. On chart review, 149 patients (33%) received an antibiotic, of whom 126 had a positive rapid strep result. Thus, based on chart review, 23 subjects (5%) diagnosed with pharyngitis received antibiotics inappropriately. Amoxicillin or penicillin was prescribed for 100 of the 126 children (79%) with a positive rapid strep test. Of the 126 children with a positive test, 114 (90%) received the correct antibiotic: amoxicillin, penicillin, or an appropriate alternative antibiotic due to b-lactam allergy. Duration of treatment was correct for all 126 children. Using the electronic algorithm, the proportion of inappropriate prescribing was 28 of 450 (6%). The test characteristics of the electronic algorithm (compared to gold standard chart review) for identification of inappropriate antibiotic prescribing were sensitivity (99%, 422 of 427); specificity (100%, 23 of 23); positive predictive value (82%, 23 of 28); and negative predictive value (100%, 422 of 422). Conclusions: For children with pharyngitis, an electronic algorithm for identification of inappropriate antibiotic prescribing is highly accurate. Future work should validate this approach in other settings and develop and evaluate the impact of an audit and feedback intervention based on this tool.
Funding: None
Disclosures: None
Development of an Electronic Algorithm to Target Outpatient Antimicrobial Stewardship Efforts for Acute Bronchitis
- Ebbing Lautenbach, Keith Hamilton, Robert Grundmeier, Melinda Neuhauser, Lauri Hicks, Anne Jaskowiak, Leigh Cressman, Tony James, Jacqueline Omorogbe, Nicole Frager, Muida Menon, Ellen Kratz, Jeffrey Gerber
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s32
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Antibiotic resistance has increased at alarming rates, driven predominantly by antibiotic overuse. Although most antibiotic use occurs in outpatients, antimicrobial stewardship programs have primarily focused on inpatient settings. A major challenge for outpatient stewardship is the lack of accurate and accessible electronic data to target interventions. We sought to develop and validate an electronic algorithm to identify inappropriate antibiotic use for outpatients with acute bronchitis. Methods: This study was conducted within the University of Pennsylvania Health System (UPHS). We used ICD-10 diagnostic codes to identify encounters for acute bronchitis at any outpatient UPHS practice between March 15, 2017, and March 14, 2018. Exclusion criteria included underlying immunocompromising condition, other comorbidity influencing the need for antibiotics (eg, emphysema), or ICD-10 code at the same visit for a concurrent infection (eg, sinusitis). We randomly selected 300 (150 from academic practices and 150 from nonacademic practices) eligible subjects for detailed chart abstraction that assessed patient demographics and practice and prescriber characteristics. Appropriateness of antibiotic use based on chart review served as the gold standard for assessment of the electronic algorithm. Because antibiotic use is not indicated for this study population, appropriateness was assessed based upon whether an antibiotic was prescribed or not. Results: Of 300 subjects, median age was 61 years (interquartile range, 50–68), 62% were women, 74% were seen in internal medicine (vs family medicine) practices, and 75% were seen by a physician (vs an advanced practice provider). On chart review, 167 (56%) subjects received an antibiotic. Of these subjects, 1 had documented concern for pertussis and 4 had excluding conditions for which there were no ICD-10 codes. One received an antibiotic prescription for a planned dental procedure. Thus, based on chart review, 161 (54%) subjects received antibiotics inappropriately. Using the electronic algorithm based on diagnostic codes, underlying and concurrent conditions, and prescribing data, the number of subjects with inappropriate prescribing was 170 (56%) because 3 subjects had antibiotic prescribing not noted based on chart review. The test characteristics of the electronic algorithm (compared to gold standard chart review) for identification of inappropriate antibiotic prescribing were the following: sensitivity, 100% (161 of 161); specificity, 94% (130 of 139); positive predictive value, 95% (161 of 170); and negative predictive value, 100% (130 of 130). Conclusions: For outpatients with acute bronchitis, an electronic algorithm for identification of inappropriate antibiotic prescribing is highly accurate. This algorithm could be used to efficiently assess prescribing among practices and individual clinicians. The impact of interventions based on this algorithm should be tested in future studies.
Funding: None
Disclosures: None





